- What Are Some Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique
- Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Instructions
- Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Chart
- Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Pdf
- Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Form
- Advantages Of Serial Dilution-agar Plate Procedure
What is Streak Plate Method ?
Streaking is a method that isolates a pure strain from a species of bacteria. A sample is taken from a colony and a microbiological culture is grown on the new plate in order for the organism to be identified properly.
What are the advantages of the serial dilution agar plate procedure Advantage: the cell count represents viable cells. Disadvantage: the method requires an incubation periods so it takes longer to get results. Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique. 10/8/2019 The Quadrajet (Q-Jet) carburetor is better on a street/daily driven vehicle for a couple reasons. One is that the primary throttle bores are pretty small, so at part throttle, like most driving, you won't be using the entire carb, and hence you'll use less gas. Serial dilutions & 'spread plate technique'? If you were to directly take a given serial dilution and do a count under a microscope what would be the advantages and disadvantages of this method versus carrying out the serial dilution-agar plate procedure to count the number of 'cells'? 1 ml of the 10-1 dilution is transferred to 9 ml sterilised saline in another test tube. This gives 100 times dilution (dilution =10-2).From the 10-2 dilution, 1 ml is dropped into a sterilised petri dish and 0.1 ml onto an agar plate, from the same pipette. For each dilution a separate sterilised pipette is used. After use it is dipped in the dispose jar.
It was Loeffler and Gaffky who first developed this method in Koch’s laboratory.
The procedure involves diluting bacteria by streaking the bacteria over the surface of the agar in the Petri dish. That way, an isolated colony can be obtained and grow into a number of cells. The culture is called a microbiological culture if the organism grows in the agar surface. (1, 2, and 3)
Picture 1: The image shows how a streak plate method is done. Pipenet free cracked.
Image Source: labmonk.com
Picture 2: The streak plate isolation method as described on the steps mentioned above.
Image Source: slideteam.net
What is the objective of the streak plate method?
The objective of streak plate method is to obtain isolated microbial colonies by creating areas of dilution on the agar petri plate. (3
What is the principle of the streak plate method?
The streak plate method requires the number of organisms in the inoculums be reduced. The procedure includes a dilution technique which requires spreading a loopful of culture over the agar plate surface.
This is to make sure that the individual cells fall apart on the agar medium surface so as separation of the different species takes place. This procedure is also called rapid qualitative isolation method. (2, 3, and 4)
Picture 3: Inoculating a plate using a streak plate technique.
Image Source: slideplayer.com
Picture 4: A pure bacterial isolate using the streak plate technique.
Image Source: researchgate.net
Picture 5: The actual result of a streak plate technique.
Image Source:slideplayer.com
What is the equipment needed for the streak plate method?
- Inoculation loop (6)
- Bacteria source
- Bunsen burner
- Striker/lighter
- Agar plate (5)
- Lysol
- Paper towel
What to keep in mind when doing a streak plate method?
- Make sure you use only a small amount of inoculum.
- Make sure you streak lightly so as not to gouge the agar.
- See to it that the plate’s surface is free of droplets of condensed moisture.
- After streaking each quadrant, do not forget to flame the loop.
- The inoculum source can be a broth or solid culture, an environmental swab, a clinical specimen, or sedimented urine.
- The petri dish to be used should be a hundred millimeter in diameter. (3, 6, 7, and 8)
What is the purpose of the streak plate technique?
The purpose of the streak plate method is to produce an isolated colony of an organism on the agar plate. Isolation of the organism is a must in a mixed culture, especially if you need to thoroughly study the colony morphology of a particular organism. (4, 7)
How is the streak plate method done?
- The inoculating loop should be sterilized in the Bunsen burner by simply putting the loop into the flame. Wait for the loop to turn red indicating that it is already hot. Let it cool down for a few minutes.
- Using the inoculating loop, pick an isolated colony from the agar plate and spread on the first quadrant of the petri plate. (4, 5)
- Gently streak the inoculating loop using a back and forth motion.
- Put the loop in the flame and let it cool. Extend the streaks into the second quarter of the petri plate.
- Repeat step four and this time extend the streak into the petri plate’s third quadrant.
- Repeat step four and go back to the area you streaked in the third quadrant of the petri plate and this time extend the streak into the fourth quadrant of the petri plate.
- Flame the loop. (8, 9, and 10)
Interpreting results
The streaked plate should be incubated for a total of 24 hours at a temperature of 37 degree Celsius. Carefully examine the colonies grown on the petri plate. The expected result is that all colonies must have the same general appearance.
If you notice that there is more than one type of colony, then you should start to streak again but this time on a separate plate so as to obtain a pure culture. (4, 5, and 6)
Streak plate method advantages and disadvantages
Advantages
- The streak plate method enables you to select and work with individual colonies.
- It is the ideal method if you are doing general work with a certain type of microorganism.
- Through the streak plate method, you will end up with a genetically identical individual colony, which makes it easy for you to grab and transfer colonies for a microscopic examination.
- The streak plate method is convenient and hassle-free as you will be able to get visible individual colonies in a single petri plate for as long as you have a good streaking skill. (2, 6, 9, and 10)
Disadvantages
- The streak plate method does not work with high volumes of organisms. It will not enable you to get a concentration count.
- It requires huge storage space and there is a possibility that your incubator cannot accommodate a large volume of petri plate.
- You will be required to prepare the agar ahead of time. The procedure can be a tedious process, especially if you don’t know the sample size before the lab work.
- Training and technique are required as various growth media have various densities to the agar. If you are not skilled enough, then you would end up tearing through the agar, especially if you apply too much pressure on the agar. A wrong streaking method can ruin your plate.
- The streak plate method can be time-consuming, especially if you are going to prepare a large sample size.
- It requires strict maintenance. The streak plate method will require constant use of the streaking loop. Eventually, it will wear out and you need to change it every once in a while. (2, 5, 7, and 10)
References
Related Posts:
Total Plate Count (TPC):
To enumerate bacteria present in a sample by serial dilution agar plating method or total plate count (TPC) method.
Purpose:
The extent of bacterial activity in a given sample in a definite set of conditions mainly depends on the total number of bacteria present in it irrespective of their species.
ADVERTISEMENTS:
Therefore, it is very often required to find out the total number of bacteria present in samples of food, water, soil, air and tissue during their microbiological analysis. This total number of bacteria includes both living and dead bacteria.’ Dead bacteria cannot grow and reproduce.
It is only the living bacteria (viable bacteria), which can grow and multiply resulting in specific bacterial activity. Therefore, it is very often required to enumerate the viable bacteria cells in different samples. However, most of the enumeration methods like direct microscopic count, electronic cell count, chemical methods and spectrophotometric method count both living as well as dead cells.
What Are Some Advantages And Disadvantages Of The Serial Dilution Agar Plate Technique
These methods cannot discriminate between living and dead cells. Therefore, serial dilution-agar plating method, which enumerates only the viable bacteria cells, is the universally used method for counting living viable cells in different samples.
Principle:
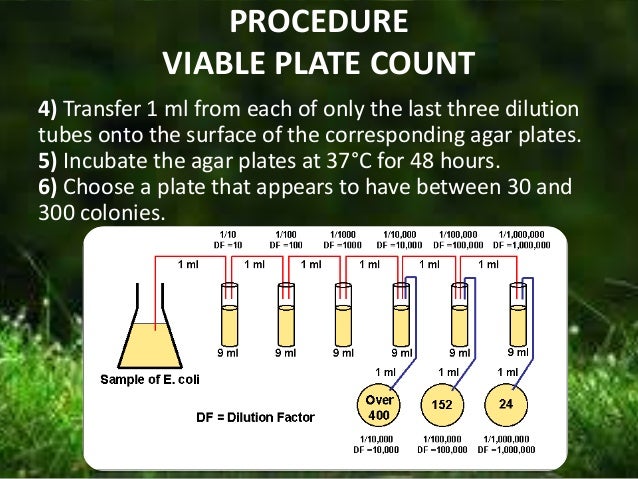
A definite weight of solid sample is homogenised aseptically in nine volumes of sterile saline to get a homogenous suspension of bacteria. The liquid sample is directly used as homogenous suspension of bacteria. The suspensions of bacteria so obtained are diluted serially (10 times, 100 times, 1000 times etc.). Here 10-1, 10-2, 10-3 etc. are called dilutions.
Their reciprocals (101, 102, 103 etc.) are called dilution factors. A definite volume of the suspension of bacteria from each dilution is inoculated onto agar plates and spread properly, so as to space the individual bacteria cells wide apart and isolate them from each other.
The inoculation of bacteria for its enumeration is done in two techniques as follows:
1. Pour plate technique
2. Spread plate technique
ADVERTISEMENTS:
1. Pour Plate Technique:
In this technique, 1 ml of the bacteria suspension is dropped onto a sterilised petri dish and then liquefied nutrient agar medium is poured over it. The petri dish is swirled gently, so as to allow the suspension to mix with the medium uniformly. It is allowed to cool and solidify.
2. Spread Plate Technique:
In this technique 0.1 ml of the bacteria suspension is dropped onto a prepared agar plate. Then, the drop of suspension is spread uniformly on the agar plate by a sterilised glass spreader.
To minimise error, each diluted suspension is plated onto 2-5 replicate plates. The inoculated plates are incubated at 37°C for 24 hours. During this period, each isolated individual bacteria cell on the agar plate grows and multiplies rapidly to produce a macroscopic visible mass of bacteria cells called a ‘colony’. Thus, the number of colonies on the plate represents the number of bacteria in the sample.
However, very often, during spreading, some cells may not get separated properly and few such unseparated cells may give rise to a single colony. Moreover, few cells have tendency to remain in pairs, chains or clusters.
Here, each pair, chain or cluster produces a colony. Thus, each colony, in strict sense, does not represent a single bacterium. That is why, instead of expressing the counts of bacteria as ‘No. of bacteria/gm or ml of sample’, it is very often expressed as number of colony forming units per gm or ml (CFU/gm or ml).
The total plate count (TPC) in the original sample is calculated by multiplying the number of CPUs with the respective dilution factors. The ‘rules of enumeration’ are followed, while calculating the number of bacteria in the original sample.
Materials Required:
ADVERTISEMENTS:
Petri dishes (15 nos.), 2-ml pipettes (10 nos.), 10-ml pipette (1 no.), test tubes (10 nos.), conical flasks (500 ml and 1 liter-1 no. each), 500 ml beaker (2 nos.), glass spreader, stainless steel pipette case, craft paper, thread (or rubber band), non-absorbent cotton, ethyl alcohol, sodium chloride (NaCl), 0.1N hydrochloric acid (HCI), 0.1N sodium hydroxide (NaOH), distilled water, nutrient agar, liquid sample (e.g. pond water/sewage water), solid sample (e.g. soil/fish meat/oyster meat/processed food), pH paper (or pH meter), pestle and mortar (or homogeniser), bunsen burner, hot air oven, autoclave, incubator, laminar flow chamber, Quebec colony counter.
Procedure:
1. Ten pipettes (in a stainless steel pipette case), 15 petri dishes and a pair of pestle and mortar (or one homogeniser cup) are sterilised in hot air oven at 180° C for 3 hours. Alternatively, they can be covered with craft paper, tied with thread or rubber band and sterilised in autoclave along with the medium (Figure 6.6).
The number of petri dishes and accordingly the amount of medium to be used is calculated depending on the number of replications and dilutions required. Here, the glassware’s and the medium have been taken for single replication and dilution upto 10-6. The number of glasswares and the amount of medium taken for sterilisation is slightly more to avoid any incidental error, because sterilisation is a lengthy process.
Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Instructions
2. 4.25 g of NaCl is dissolved in 500 ml of distilled water to get physiological saline (0.85%). 225 ml of this saline solution is poured into a 500 ml conical flask. Iptv tool for mac. Its mouth is cotton-plugged, covered with craft paper and tied with thread or rubber band. 18 wos haulin. It is used as the first diluents to dilute the solid sample.
3. 9.0 ml of the left over saline is also pipetted into each of the 10 test tubes. Their mouths are cotton-plugged, covered with craft paper and tied with thread or rubber band. These are used as diluents for serial dilution.
4. The ingredients of nutrient agar medium or its ready-made powder required for 500 ml of the medium is weighed and dissolved in 500 ml of distilled water in a 1 liter conical flask by shaking and swirling.
Its pH is determined using a pH paper or pH meter and adjusted to 7.0 using 0.1N HC1 if it is more or using 0.1N NaOH if it is less. The flask is heated to dissolve the agar in the medium completely. Then, it is cotton-plugged, covered with craft paper and tied with thread or rubber band.
5. The 500 ml conical flask containing 225 ml of saline, the 10 test tubes containing 9 ml of saline each and the 1 liter conical flask containing 500 ml of nutrient agar medium are sterilized at 121°C (15 psi pressure) for 15 minutes in an autoclave.
Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Chart
6. After sterilisation, the sterilised materials are removed from the autoclave and allowed to cool for some time, without allowing the medium to solidify. Cooling of the medium prevents condensation and accumulation of water droplets inside the plates. If the medium has already been prepared and solidified during storage, it has to be liquefied by heating carefully till it melts completely.
7. To prepare agar plates, before the sterilised nutrient agar medium cools and solidifies, in warm molten condition, it is poured aseptically into the 6 sterilised petri dishes (approximately 20 ml each), so that the molten medium covers the bottom of the petri dishes completely.
Then, the plates are covered with their lids and allowed to cool, so as to solidify the medium in them. Water vapour that may condense on the inner surface of the plates and lids is evaporated by keeping the plates and lids in inverted position in an incubator at 37°C for about 1 hour.
8. 25 g of the solid sample (e.g. fish meat/oyster meat/processed food) is weight and homogenised in 225 ml sterilised saline (diluent) aseptically (Figure 6.7). This gives a 10 times dilution (dilution = 10-1). For the liquid sample, 1ml of sample is pipetted aseptically into a 9 ml sterilised saline tube. This also gives a 10 times dilution (dilution = 10-1).
9. 1 ml of the 10-1 dilution is transferred to 9 ml sterilised saline in another test tube. This gives 100 times dilution (dilution =10-2). From the 10-2 dilution, 1 ml is dropped into a sterilised petri dish and 0.1 ml onto an agar plate, from the same pipette. For each dilution a separate sterilised pipette is used. After use it is dipped in the dispose jar.
10. 1 ml of the 10-2 dilution is transferred to 9 ml sterilised saline in another test tube. This gives 1000 times dilution (dilution =10-3). From the 10-3 dilution, 1 ml is dropped into a sterilised petri dish and 0.1 ml onto an agar plate, from the same pipette. In a similar way, dilution is continued upto 10-6 serially, each time transferring 1ml to a sterilised petri dish and 0.1 ml to an agar plate from the same pipette.
11. Then, the drops of suspension on the agar plates are spread aseptically by a sterilised glass spreader. After spreading in each plate, it is flame-sterilised by dipping in alcohol and showing over a flame. This is the ‘spread plate technique’.
12. The petri dishes containing 1ml of bacteria suspension each are taken and sterilised liquefied nutrient agar is poured into them. They are swirled gently, so as to allow the suspension to mix with the medium uniformly. Photoshop cc 2017 full crack for mac from torrent. The plates are allowed to cool till the medium solidifies. This is ‘pour plate technique’.
ADVERTISEMENTS:
13. Then, the plates are incubated in inverted position, top down, at 37°C for 24 hours in an incubator (Figure 6.7).
14. An un-inoculated agar plate is incubated as control to ensure proper sterilisation as shown by no growth on it.
Observations:
The number of colonies of bacteria on the plates is counted directly or with the help of a Quebec colony counter. From this, the number of bacteria present per gram or ml of the original sample is calculated. This is called enumeration.
Rules of Enumeration:
1. Petri dishes with 30 to 300 colonies should be considered.
2. Average numbers of duplicates and triplicates (R1, R2 R3…) are considered only if one count is not more than double of the other. If one is more than double of the other lower value is taken. Acoustic solutions lcdw19hdf manual.
Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Pdf
3. For pour plate technique, bacterial count is No.X 10c/gm, where c = dilution factor. For spread plate technique, bacterial count is No. X10c+1/gm, where c = dilution factor. The number is converted to two decimal places in the form of (x.yz X 10m). For example, 288 X 104 is expressed as 2.88 X 106.
4. If, in all dilutions, colony number is more than 300, count for highest dilution and if in all dilutions, it is less than 30, count for the lowest dilution is considered. In both cases count is represented as: Estimated No. X 10c/gm or ml for pour plate technique and Estimated No. X 10c+1/gm or ml for spread plate technique.
Advantage And Disadvantage Of The Serial Dilution Agar Plate Procedure Form
5. If no colony is observed in any dilution taken, it is represented as: Estimated <1 x lowest dilution.
Advantages Of Serial Dilution-agar Plate Procedure
6. As the serial dilution takes place in terms of 10 times, mathematically it is obvious that no two dilutions can have colonies between 30 and 300. For example, if 10-3 has 50 colonies, 10-2 should have 500 (i.e. >300) and 10-4 should have 5 (i.e. <30) colonies.
However, this does not occur in reality, as bacteria do not occur as a homogenous solution; rather occur as a suspension in the diluents. If there are two dilutions having countable colonies (between 30 and 300) first calculate the number of colony forming units/gm or ml using each dilution.
If one value is more than double of the other, report the lower value. If not, take the average of the two values and report that value.
